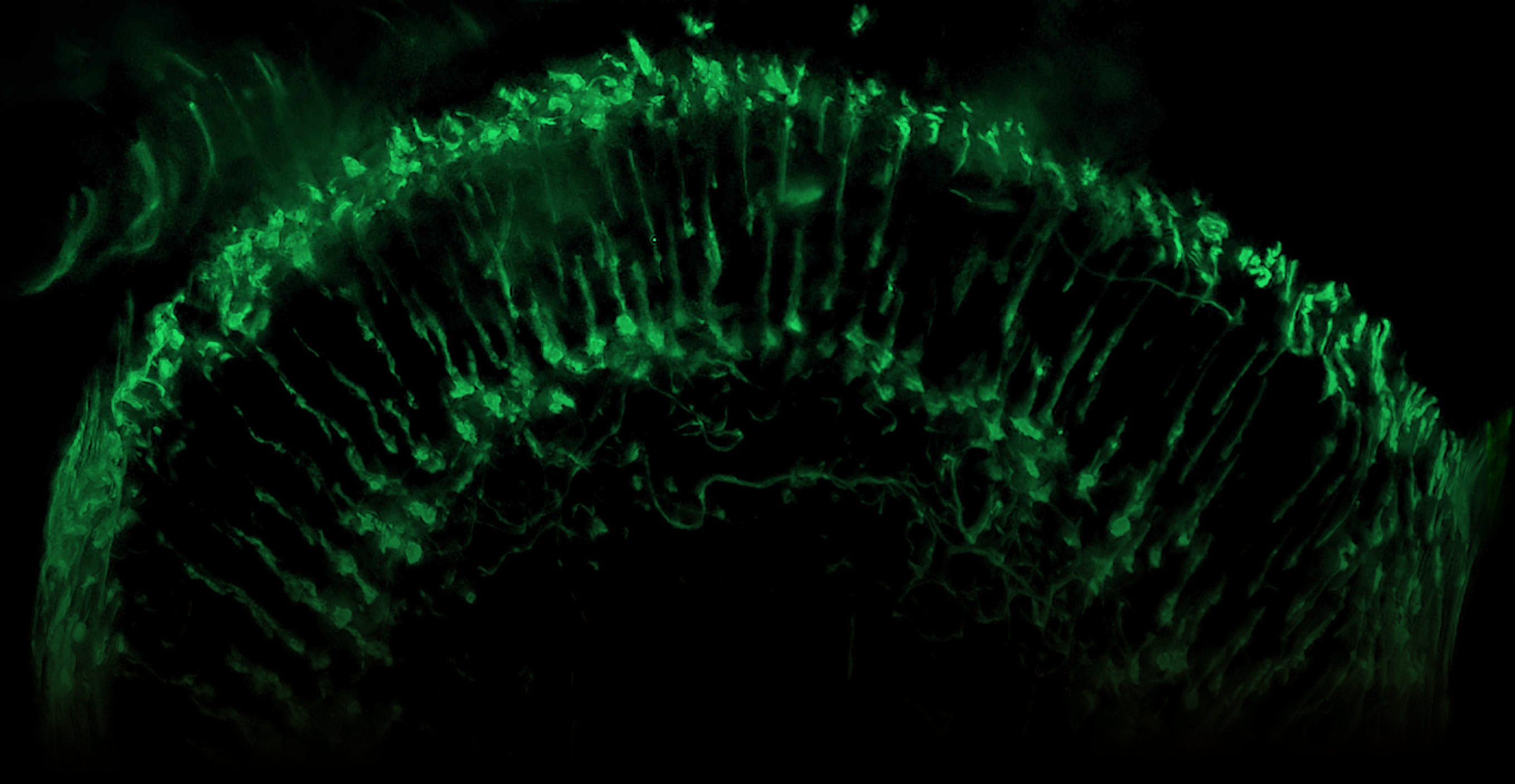
What We Do
We Study Vision
At O'Tousa Lab, we combine a wide range of molecular, biochemical, and cellular methods to analyze genes responsible for normal visual function.
Drosophila
With over 40 years of study by dozens of labs, Drosophila melanogaster is the premiere research organism for the study of vision and light-related responses. Its eyes are well suited for observing molecular pathways and cell-to-cell communication systems underlying key development processes.
Mosquitoes
The ability to do genome editing now allows our molecular genetic approaches to be applied to mosquito research. We characterize photoreceptor specializations allowing mosquitoes to acquire visual information in very low light environments. The ability of mosquitoes to integrate this information with other sensory inputs is the basis of efficient host seeking and other specialized mosquito behaviors.

Drosophila Vision
Expertise with the genetic approaches available in Drosophila has allowed my laboratory to use this invertebrate model to study the process of vision and retinal degeneration. My research contributed to the identification of the first invertebrate rhodopsin, showing that this light sensor is an evolutionary relative of vertebrate rhodopsins. We determined that missense mutations in Drosophila rhodopsin are responsible for retinal degeneration, a finding correctly predicting that rhodopsin mutations can cause retinal degenerations in humans. We further showed hyperphosphorylation of rhodopsin is a trigger of retinal degeneration while carrying out the molecular characterization of other genes required for rhodopsin maintenance and the prevention of retinal degeneration. While the focus of my research group has been on the Drosophila system, we contributed insights into the retinal organization of mosquito visual system. The rhodopsin biology of these dim light and nocturnal organisms is much different than Drosophila and presents an excellent model to study light-driven physiology of high sensitivity photoreceptors.

Mosquito Vision
Rhodopsins are the G-protein coupled receptors initiating visual transduction. Animal genomes typically contain multiple rhodopsin genes with different spectral properties such that the expression of different rhodopsins in distinct classes of photoreceptor cells provides the basis for color vision. There are 10 predicted rhodopsin genes in both Ae. aegypti and An. gambiae genomes. These are classified into five different groups on the basis of comparison to Drosophila and other invertebrate rhodopsins. These are a long wavelength group (most sensitive to to blue/green light) of five members, the short wavelength (most sensitive to UV/blue light) Aaop9, the UV sensitive Aaop8, and the two poorly characterized groups represented by Aaop10 and the pteropsin Aaop12. This large family of rhodopsins is also present in Culex genomes, suggesting a common use of visual information in the disease-related mosquitoes.
The identification of the photoreceptor cell type expressing each of these rhodopsins is key to understanding the organization of the mosquito retina and the mechanisms involved in the processing of visual information. In the Drosophila retina, there are two major classes of ommatidia distinguished by the pairing of rhodopsin expression in the R7 and R8 cell types. We recently showed that the UV sensitive Aaop8 rhodopsin and a long wavelength sensitive Aaop2-type rhodopsin are expressed in non-overlapping subsets of the Ae. aegypti R7 photoreceptor cells. We followed this work with a second report showing that other rhodopsins are coexpressed in these R7 cells. These studies as well as other unpublished work in the laboratory show the overall pattern of rhodopsin expression in mosquitoes is not as predicted from the Drosophila model. We will use this information to determine the basis of visually mediated behaviors of these insects.
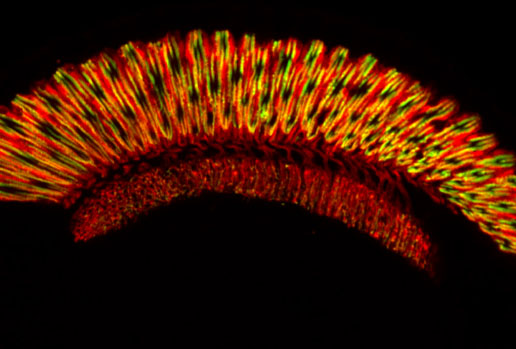
Genetic Triggers of Retinal Degeneration
Understanding the molecular events and the proteins involved in the regulation of visual process provides insights into the basic operation of cellular signaling which in turn allows the development of methodologies to treat retinal degeneration and other visual system disorders. The molecular characterization of human, other vertebrate, and Drosophila retinal degeneration syndromes has revealed that defects in many distinct biochemical pathways trigger degeneration. We are using genetic techniques to identify the molecular components of the cell death pathways existing in adult photoreceptors.
Light-driven posttranslational modifications of rhodopsin play a critical role in both light-evoked photoreceptor response and the maintenance of photoreceptor cell health. These dual roles are observed in both vertebrate and invertebrate photoreceptors, and aberrant activity will trigger retinal degeneration. We are studying the role of the light-driven binding of arrestin in controlling two key posttranslational modifications of Drosophila Rh1: phosphorylation and ubiquitination. The interplay of these processes control the rate of rhodopsin regeneration and rhodopsin internalization, to promote photoreceptor viability.

Recent Publications
Rocha, M., Kimler, K.J., Leming, M.T., Hu, X., Whaley, M.A., and O'Tousa, J.E., (2015) Expression and light-triggered movement of rhodopsins in the larval visual system of mosquitoes. J Exp Biol. 218, 1386-1392.
Mecklenburg, K.L., Freed, S.A., Raval, M., Quintero, OA, Yengo, C.M., and O'Tousa, J.E. (2015) Invertebrate and vertebrate class III myosins interact with MORN repeat-containing adaptor proteins, PLoS ONE 10(3): e0122502. doi:10.1371/journal pone.0122502.
Champion, M.M., Sheppard, A.D., Rund, S.C., Freed, S.A., O’Tousa,J.E., and Duffield, G.E. (2016) Qualitative and Quantitative Proteomics Methods for the Analysis of the Anopheles gambiae Mosquito Proteome. In:Short Views on Insect Genomics and Proteomics (ed: C. Raman, M.Goldsmith, T. Agunbiade) Springer, p. 37-62.
Metoxen, A.J., Leming, M.T., Hu, X., Whaley, M.A., and O'Tousa, J.E.(2016) Light-driven processes control both rhodopsin maturation and recycling in mosquito photoreceptors. J. Neurosci. 36, 11051-11058.[This report was recommended as being of special significance in its field by the F1000 Faculty: (http://f1000.com/prime/726906631?subscriptioncode=fa7fb20b-1c27-45f5-bce2-53322a7c7933).]
Mecklenburg, K.L., Weghorst P.L., Freed, S.A., O'Tousa, J.E. (2018) Discordant responses to MAPK pathway stimulation include axonal growths in adult Drosophila photoreceptors. Frontiers Mol. Neuro. 11, p. 441-452.
Tang, J.Y., Kosgei, J., Ochomo, E., Ndenga, B.A., Ghiaseddin, R., Lobo, N.F., Hawkes, F.M. and O'Tousa, J.E. (2020), Impact of visual features on capture of Aedes aegypti with host decoy traps (HDT). Med Vet Entomol. https://doi.org/10.1111/mve.12482.

Who We Are
| Joe O'Tousa | Lab Head |
| Michelle Whaley | Faculty |
| Kirk Mecklenburg | Visiting Scientist |
| Matt Gregory | Research Technician |
| Aidan Cook | Undergraduate Researcher |
| Hannah Cunniff | Undergraduate Researcher |
| Brian Dineen | Undergraduate Researcher |
| Miguel Girod | Undergraduate Researcher |
| Patrick Kollman | Undergraduate Researcher |
| Calla Sullivan | Undergraduate Researcher |
| Elaine Teeters | Undergraduate Researcher |