Multimedia Publications
*Slideshare webinars and videos of research from our group
Videos
Follow us on YouTube for all of our latest webinars and videos.
Watch Jeff DuBose's Lecture Video on Instrumentation for Nanosecond and Femtosecond Transient Absorption Spectroscopy:
Watch Jeff DuBose's Lecture Video on Principles of Transient Absorption Spectroscopy:
Watch Jeff DuBose's symposium talk on Directing Energy Transfer in Halide Perovskite-Chromophore Hybrid Assemblies :
Watch Prashant's Webinar on the PhD and Beyond. Laying the Foundation for a Successful Career:
Watch Jeff DuBose's lecture on Time Correlated Single Photon Counting (TCSPC) for Photoluminescence Lifetime Measurements:
Preethi Mathew's nanoGe poster presentation on Iodide Expulsion in Perovskites:
Jeff DuBose's nanoGe poster presentation on TiO2-Assisted Halide Ion Segregation:
Undergraduate Researcher Jake Drysdale Gives Poster Presentation about his project:
Halide Composition and Temperature Dependent Properties of Perovskite Solar Cells:
Dr. Kamat's Webinar on Light Energy Harvesting (Sigma Aldrich)
How do semiconductor nanostructures offer new opportunities to tackle clean energy challenge? What are different ways with which we can design light harvesting assemblies for conversion of light energy into electrical energy or chemical energy? What is perovskite photovoltaics and how it can compete with the current photovoltaic technology? This webinar aims to address these questions.
Measuring Photoelectrochemical Performance of QDSCs - Interpreting 2 and 3 Electrode Measurements Transformation of CH3NH3PbI3 in Humid Air Perovskite Solar Cells - C&EN Story Making High Efficiency Quantum Dot Solar Cells Prashant Kamat - ND Thinks Big Painting Solar Cells with Nanoparticle Paste SrTiO3 for Water Splitting Graphene for Energy Conversion Graphene-TiO2 nanocomposites Electrophoretic DepositionA Donut Solar Cell:
Webinars
- Perovskite Photocatalysis. Methyl Viologen Induces Unusually Long-Lived Charge Carrier Separation in CsPbBr3 Nanocrystals
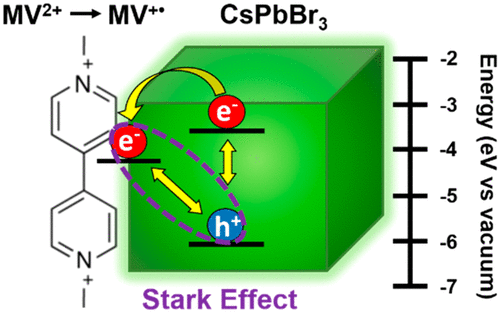
Jeff DuBose (graduate student and first author of paper) describes the unique excited state interaction between CsPbBr3 perovskite nanocrystals and methyl viologen - a molecular electron shuttle.
535. Perovskite Photocatalysis. Methyl Viologen Induces Unusually Long-Lived Charge Carrier Separation in CsPbBr3 Nanocrystals Steven M. Kobosko, Jeffrey T. DuBose and Prashant V. Kamat . ACS Energy Lett. 2020
- Interfacial Charge Transfer between Excited CsPbBr3 Nanocrystals and TiO2: Charge Injection versus Photodegradation
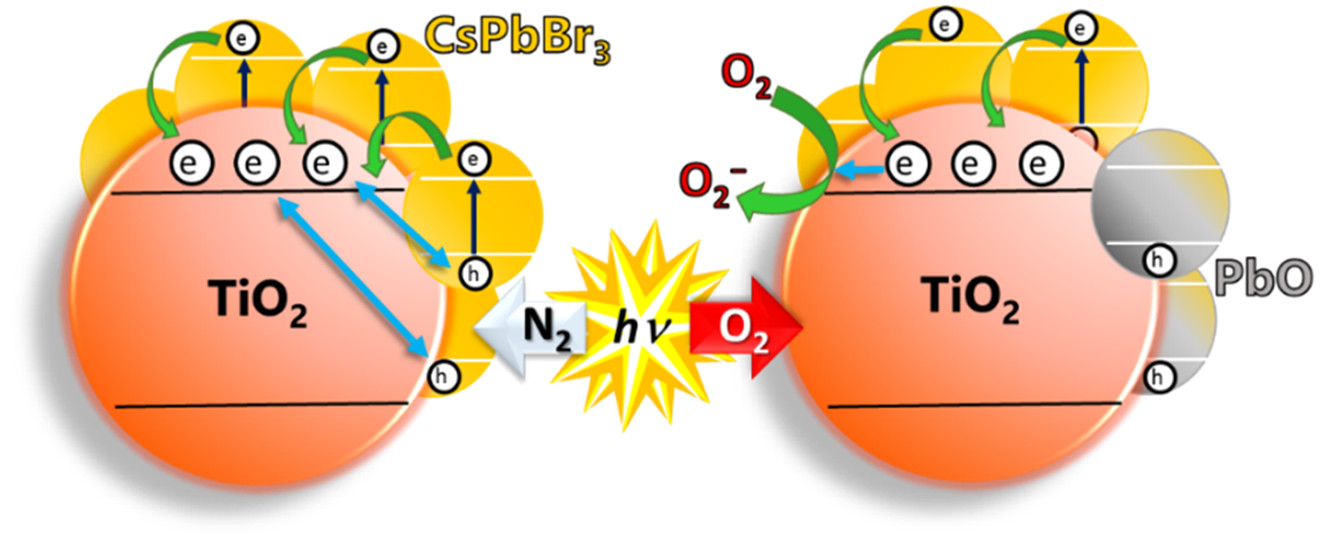
Rebecca Scheidt - third year grad student and first author of this paper - discusses the effect of different metal oxide surfaces on the excited state of perovskite quantum dots via a LiveSlides presentation!
521. Interfacial Charge Transfer between Excited CsPbBr3 Nanocrystals and TiO2: Charge Injection versus Photodegradation Rebecca A. Scheidt, Elisabeth Kerns, and Prashant V. Kamat. J. Phys. Chem. Lett. 2018, 9, 20, 5962-5969
- CdSeS Nanowires. Compositionally Controlled Band Gap and Exciton Dynamics

Jeff Christians discusses a simple solution-based synthesis developed in the Kamat Lab which allows for the synthesis of highly crystalline CdSeS nanowires. Tuning of the composition by the ratio of Se:S precursors allows for the modulation of the NW band gap between 2.36 eV and 1.79 eV. Thus, these NWs have great promise for incorporation into optoelectronic devices.
457. CdSeS Nanowires. Compositionally Controlled Band Gap and Exciton Dynamics Kim J.-P.; Christians, J. A.; Choi, H.; Krishnamurthy, S.; Kamat, P. V. J. Phys. Chem. Lett. 5, 1103-1109.
- Rate Limiting Interfacial Hole Transfer in Sb2S3 Solid-State Solar Cells
Jeff Christians discusses the importance of hole transfer to the performance of Sb2S3 solar cells, specifically highlighting the mechanism of hole transfer in these solid-state solar cells and discriminating between the processes of hole diffusion in the Sb2S3 layer and hole transfer across the Sb2S3 interface.
451. Rate Limiting Interfacial Hole Transfer in Sb2S3 Solid-State Solar Cells Christians, J. A.; Leighton Jr., D. T.; Kamat, P. V. Energy and Environ. Sci. 2014, 7, 1148-1158.
- An Inorganic Hole Conductor for Organo-Lead Halide Perovskite Solar Cells. Improved Hole Conductivity with Copper Iodide

Jeff Christians discusses CuI and its promising use as an inorganic hole conductor that can be deposited from solution while still being compatible with organo-metal lead halide perovskite. Copper iodide offers improved stability and higher electrical conductivity than the commonly used spiro-OMeTAD.
448. An Inorganic Hole Conductor for Organo-Lead Halide Perovskite Solar Cells. Improved Hole Conductivity with Copper Iodide Christians, J. A.; Fung, R. C. A.; Kamat, P. V. J. Am. Chem. Soc. 2014, 136 (2), 758-764.
- Trap and Transfer. Two-Step Hole Injection Across the Sb2S3/CuSCN Interface in Solid State Solar Cells.

Jeff Christians discusses the mechanism and rate of hole transfer from Sb2S3 to CuSCN. It is found that holes are initially trapped in the Sb2S3 as sulfide radical (S−•) and are then transferred to CuSCN. This highlights several important issues related to the design of Sb2S3/CuSCN solar cells and offers up potential strategies for increasing solar cell efficiency.
443. Trap and Transfer. Two-Step Hole Injection Across the Sb2S3/CuSCN Interface in Solid State Solar Cells. Christians, J. A.; Kamat, P. V. ACS Nano 2013, 7 (9), 7967–7974.
- Making Graphene Holey. Gold-Nanoparticle-Mediated Hydroxyl Radical Attack on Reduced Graphene Oxide

James Radich discusses how gold nanoparticles mediate the hydroxyl radical attack of reduced graphene oxide forming holes in the graphene structure. This raises important issues for the stability of graphene based assemblies in certain environments, but also opens up the possiblity of new graphene structures for improved devices.
439. Making Graphene Holey. Gold-Nanoparticle-Mediated Hydroxyl Radical Attack on Reduced Graphene Oxide Radich, J. G.; Kamat, P. V. ACS Nano 2013, 7 (6), 5546–5557.
- Galvanic Exchange on Reduced Graphene Oxide

Sachidananda Krishnamurthy discusses the design of new multifunctional catalyst materials by performing a galvanic exchange of metal nanoparticles on the surface of reduced graphene oxide (RGO). This synthesis technique opens up the possibility of synthesizing new metal nanoparitcle-RGO composite catalysts.
432. Galvanic Exchange on Reduced Graphene Oxide. Designing a Multifunctional Two-Dimensional Catalyst Assembly. Krishnamurthy, S.; Kamat, P. V. J. Phys. Chem. C 2013, 117 (1), 571–577.
- Tandem-Layered Quantum Dot Solar Cells

Dr. Pralay Santra gives a short review of his paper, in which he demonstrates synergistic effects in QDSSCs that employ a layered structure of CdSeS QDs with decreasing band gaps (by emission, green, yellow, red). This tandem structure boosted the efficiency of the QDSSC to 3.0%, a 60% increase compared to premixed QDs.
433. Tandem-Layered Quantum Dot Solar Cells: Tuning the Photovoltaic Response with Luminescent Ternary Cadmium Chalcogenides. Santra, P.; Kamat, P. V. J. Am. Chem. Soc. 2013, 135 (2), 877–885.
- Solar Energy - Beyond the Hype

Professor Prashant Kamat examines (1) the facts of how energy is used and created around the world, (2) how different types of carbon neutral energy sources could be used to meet growing energy demands, and (3) the ways our lab has sought to develop solar devices to meet this growing energy demand.
- Measuring Photoelectrochemical Performance of QDSCs

In this webinar, Jeff Christians looks at the photoelectrochemical techniques used to measure solar cell performance. This includes the theory and application of both 2 and 3 electrode measurements, as well some basic background information needed to properly interpret and understand these measurements.
- Origin of RGO Enhancements in Electrochemical Energy Storage

James Radich gives a short review of his paper, in which he investigates the causes behind RGO enhancements in electrochemical energy storage by probing the electrochemical response of an α-MnO2/RGO composite used as an electrode in a lithium ion battery cell and elucidating the mechanistic aspects of the RGO on the commonly observed improvements in cycling and capacity.
425. Origin of Reduced Graphene Oxide Enhancements in Electrochemical Energy Storage. Radich, J. G.; Kamat, P. V. ACS Catal. 2012, 2, 807–816.
- Reduced Graphene Oxide Composite Counter Electrode for Quantum Dot Solar Cells

James Radich details a reduced graphene oxide-Cu2S composite counter electrode that shows high redox activity toward teh polysulfide electrolyte used in many QDSCs. Using this counter electrode, a sandwich CdS/CdSe quantum dot sensitized solar cell was constructed that exhibited a 4.4% power conversion efficiency.
416. Cu2S -Reduced Graphene Oxide Composite for High Efficiency Quantum Dot Solar Cells . Overcoming the Redox Limitations of S2-/Sn2- at the Counter Electrode Radich, J. G.; Dwyer, R.; Kamat, P. V. J. Phys. Chem. Lett. 2011, 2, 2453–2460.
- Sun-Believable Solar Paint

A transformative approach to solar energy harvesting, a semiconductor based solar paint, is outlined by Ian Lightcap. This binder-free paste consisting of CdS, CdSe, and TiO2 nanoparticles was applied in a one step process to conduction glass and annealed at 473K. Showing power conversion efficiencies of greater than 1% under ambient conditions, this paint offers the advantages of simple design and low cost to the next generation of solar cells.
418. Sun-believable Solar Paint. A Transformative One-Step Approach for Designing Nanocrystalline Solar Cells Genovese, M.; Lightcap, I. V.; Kamat, P. V. ACS Nano 2012, 6 (1), 865–872.
- Charge Transfer Complexation and Excited State Interactions in Porphyrin-Ag NP Structures

Sean Murphy details the interactions in porphyrin-silver nanoparticle hybrid nanostructures. Strong Raman enhancement indicates both electromagnetic and chemical enhancement. Evidence of chemical enhancement includes a selective enhancement of porphyrin Raman bands. Fast charge separation in the complex is indicated by ultrafast transient absorption and fluorescence upconversion measurements.
413. Charge-Transfer Complexation and Excited State Interactions in Porphyrin-Silver Nanoparticle Hybrid Nanostructures Murphy, S.; Huang, L.; Kamat, P. V. J. Phys. Chem. C 2011, 115 (46), pp 22761–22769.
- Electron Transfer Between Methyl Viologen Radicals and Graphene Oxide
Graphene based nanoassemblies are interesting because of their potential in designing electronic, sensing, and energy conversion devices. Sachidananda Krishnamurthy shows in this webinar, graphene oxide's ability to accept electrons from methyl viologen which results in the reduction of GO as well as the storage of electrons. Sachi then shows how these stored electrons can be used to reduce Ag+ ions, providing an interesting assembly with anchored silver nanoparticles on the reduced graphene oxide.
406. Electron Transfer between Methyl Viologen Radicals and Graphene Oxide: Reduction, Electron Storage and Discharge Krishnamurthy, S.; Lightcap, I. V.; Kamat, P. V. J. Photochem. Photobiol. A:Chem. 2011, 221, 214-219.
- Adsorption and Electron Injection for CdSe on TiO2

Doug Pernik, an undergraduate in the Kamat lab, looks at the kinetics involved in the adsorption of quantum dot films, including both the Langmuir-like submonolayer adsorption and QD aggregation processes. Adsorption constants and electron injection rates from the QD into TiO2 were determined with the goal of controlling CdSe QD adsorption and designing efficient photoanodes for QDSCs.
408. Tracking the Adsorption and Electron Injection Rates of CdSe Quantum Dots on TiO2: Linked Versus Direct Attachment Pernik, D.; Tvrdy, K.; Radich, J. G.; Kamat, P. V. J. Phys. Chem. C 2011, 133 (24), pp 9607–9615.
- The Role of IrO2 in Mediating Hole Transfer at the TiO2 Interface

Ben Meekins shows how IrO2, a water oxidation cocatalyst, plays an important role in mediating the hole transfer process of a UV-irradiated TiO2 system. Ben gives insight into the mechanism and kinetics of the catalytic role of IrO2 in the photogenerated hole transfer process.
415. Role of Water Oxidation Catalyst, IrO2 in Shuttling Photogenerated Holes Across TiO2 Interface Meekins, B. H.; Kamat, P. V., J. Phys. Chem. Lett. 2011, 2, 2304-2310.
