Graphene Chemistry
Background
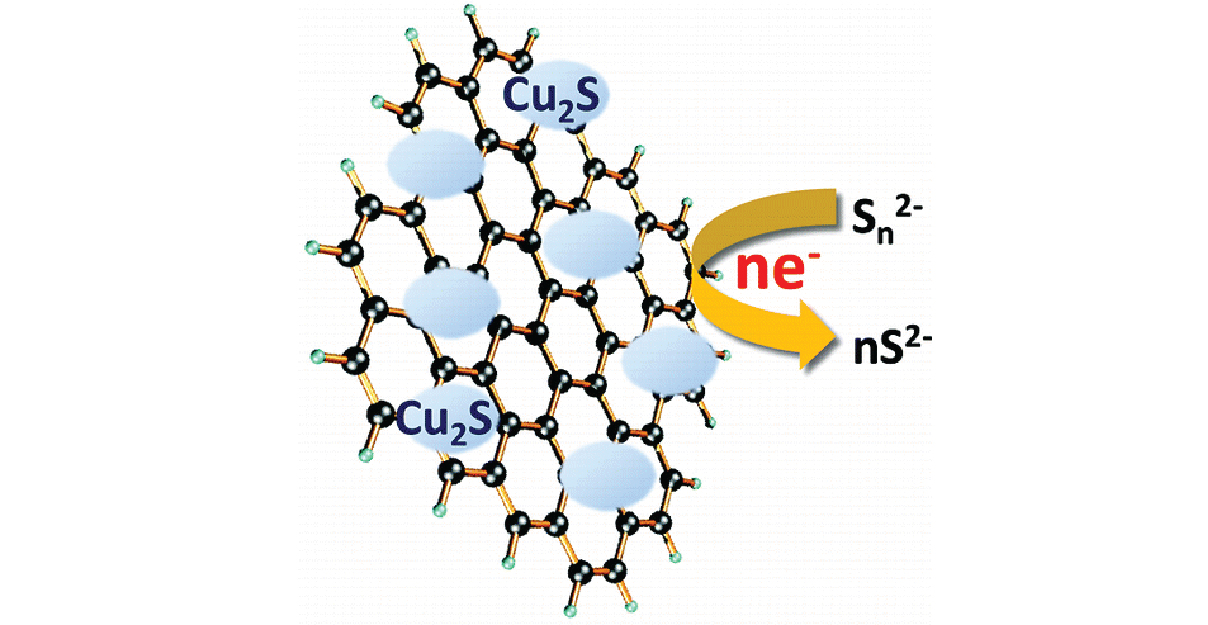
Figure. Cu2S/RGO composite developed for use as a counter electrode for quantum dot solar cells employing a sulfide/polysulfide redox couple.
Graphene just might be the best thing since sliced, gluten-free, bread. The single-layer carbon allotrope is cheap, robust, has impressive surface area, and can be readily produced through chemical oxidation in the form of graphene oxide (GO). Our research efforts focus on elucidating and harnessing graphene's versatile set of electronic interactions with semiconductors and metal nanoparticles.
The electron storage and shuttling ability of GO is a significant driving force behind the creation of graphene-metal and graphene-semiconductor-metal nanoparticle composites utilized for next-generation energy conversion and sensing devices.
Check out these links for more info!
- Making Graphene Holey. Gold-Nanoparticle-Mediated Hydroxyl Radical Attack on Reduced Graphene Oxide - Webinar on the solution based fabrication of porous graphene as a potential material for Li-ion batteries, fuel cells, and catalysis.
- Graphene-based Nanoassemblies for Energy Conversion - Video from the perspective article: Graphene-based Nanoassemblies for Energy Conversion. Kamat, P. V. J. Phys. Chem. Lett. 2011, 2, 242-251
- Origin of Enhancements in Li-ion Batteries using RGO - Webinar detailing the origin of performance enhancements in Li-ion batteries utilizing reduced graphene oxide
- Electron Transfer Between Methyl-Viologen Radicals and Graphene Oxide - Webinar showing the development of Ag/RGO nanocomposits by taking advantage of GOs ability to store and shuttle electrons
Our Research Focus
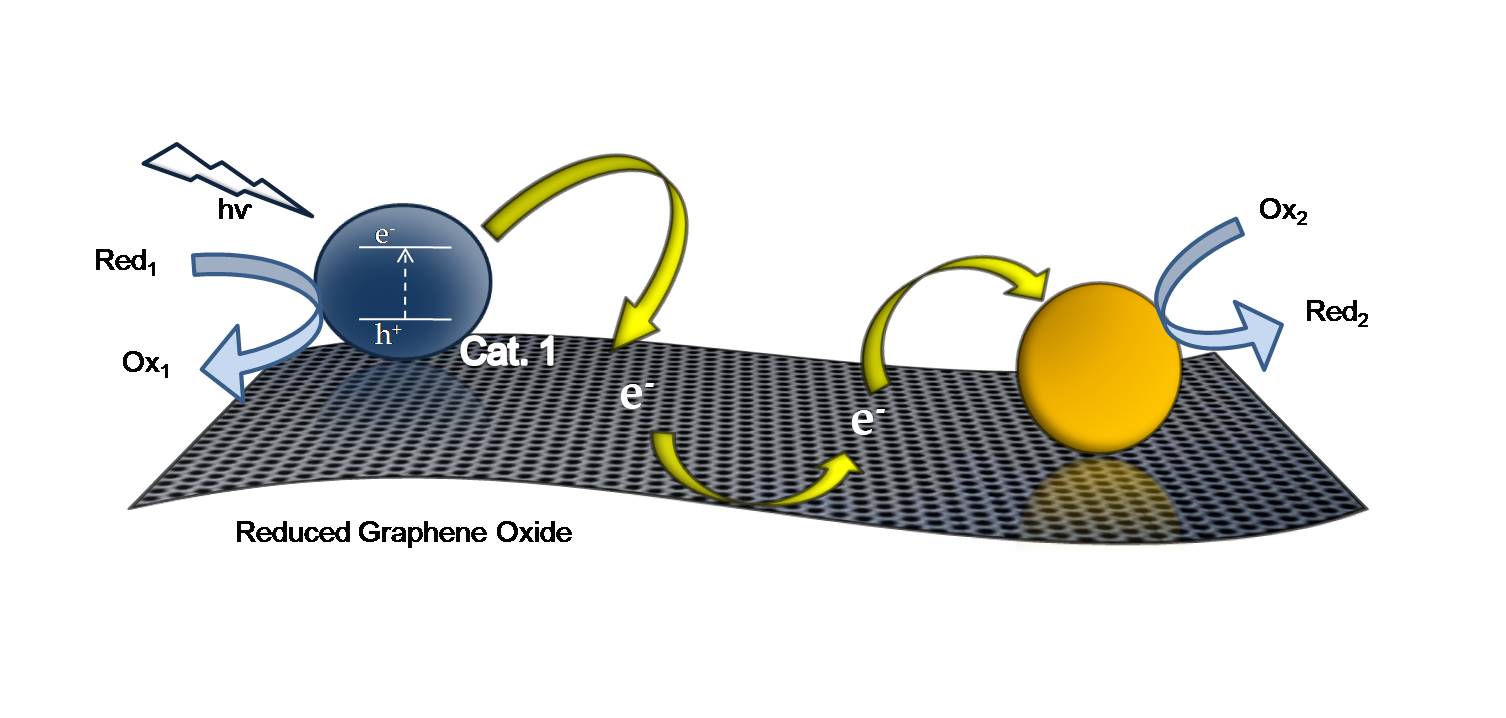
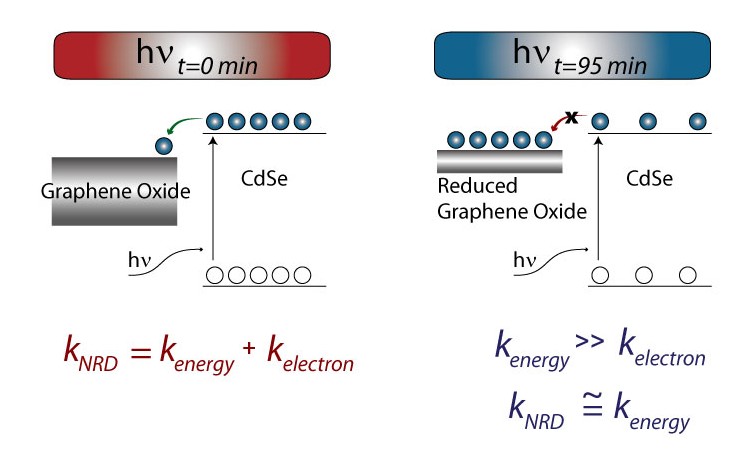
Figure. By monitoring fluorescence, electron transfer to GO can be separated from energy transfer in semiconductor-GO systems because after long illumination times the electron transfer pathway becomes negligible (left). Reduced graphene oxide (RGO) can act as a catalyst mat; it is able to store, and shuttle electrons for a photoactive semiconductor to a metal reaction center (right).
Electronic Properties of GO composites
- Electron storage and shuttling
- Charge transfer interactions between GO and semiconductors, metals, and molecular species
- Electron and energy transfer kinetics
- Electron hopping
Figure. Semidonuctor-graphene-metal (SGM) films were developed to detect nanomolar concentrations using surface enhanced raman scattering (SERS) detection.
GO Composite Devices
- Solar Fuels
- Batteries
- Quantum Dot Solar Cells
- Chemical Sensors
- Catalyst Mats
Recent Progress
- Developed a solution method for the production of porous graphene sheets
- Improved the performance of Lithium Ion batteries by the incorporation of graphene
- Fabricated multifunctional catalsts on the surface of (reduced) graphen oxide
- Developed a Cu2S/RGO composite for use as a counter electrode material in quantum dot solar cells
- Showed the ability of reduced graphene oxide to store, shuttle, and discharge electrons
Select Publications
447. Direct Observation of Spatially Heterogeneous Single-Layer Graphene Oxide Reduction Kinetics McDonald, M. P.; Eltom, A.; Vietmeyer, F.; Thapa, J.; Morozov, Y. V.; Sokolov, D. A.; Hodak, J. H.; Vinodgopal, K.; Kamat, P. V.; Kuno, M. Nano Lett. 2013, 13 (12), 5777-5784.
444. Nickel-Doped MnO2 Nanowires Anchored onto Reduced Graphene Oxide for Rapid Cycling Cathode in Lithium Ion Batteries Radich, J. G.; Chen, Y.-S.; Kamat, P. V. ECS Journal of Solid State Science and Technology 2013, 2 (10), M3178-M3181.
439. Making Graphene Holey. Gold-Nanoparticle-Mediated Hydroxyl Radical Attack on Reduced Graphene Oxide Radich, J. G.; Kamat, P. V. ACS Nano 2013, 7 (6), 5546–5557.
434. Reduced Graphene Oxide–Silver Nanoparticle Composite as an Active SERS Material. Murphy, S.; Huang, L.; Kamat, P. V. J. Phys. Chem. C 2013, 117 (9), 4740–4747.
432. Galvanic Exchange on Reduced Graphene Oxide. Designing a Multifunctional Two-Dimensional Catalyst Assembly. Krishnamurthy, S.; Kamat, P. V. J. Phys. Chem. C 2013, 117 (1), 571–577.
431. Graphitic Design: Prospects of Graphene-Based Nanocomposites for Solar Energy Conversion, Storage, and Sensing. Lightcap, I. V.; Kamat, P. V. Acc. Chem. Res. 2013, 46 (10), 2235–2243.
429. Dual-Frequency Ultrasound for Designing Two Dimensional Catalyst Surface: Reduced Graphene Oxide-Pt Composite. Vinodgopal, K.; Neppolian, B.; Salleh, N.; Lightcap, I. V.; Grieser, F.; Ashokkumar, M.; Ding, T. T.; Kamat, P. V. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2012, in press.
426. Electron Hopping Through Single-to-Few Layer Graphene Oxide Films. Photocatalytically Activated Metal Nanoparticle Deposition. Lightcap, I. V.; Murphy, S.; Schumer, T.; Kamat, P. V. J. Phys. Chem. Lett. 2012, 3, 1453–1458.
425. Origin of Reduced Graphene Oxide Enhancements in Electrochemical Energy Storage. Radich, J. G.; Kamat, P. V. ACS Catal. 2012, 2, 807–816.
424. Fortification of CdSe Quantum Dots with Graphene Oxide. Excited State Interactions and Light Energy Conversion. Lightcap, I. V.; Kamat, P. V. J. Am. Chem. Soc., 2012, 134 (16), 7109–7116.
416. Cu2S -Reduced Graphene Oxide Composite for High Efficiency Quantum Dot Solar Cells . Overcoming the Redox Limitations of S2-/Sn2- at the Counter Electrode Radich, J. G.; Dwyer, R.; Kamat, P. V. J. Phys. Chem. Lett. 2011, 2, 2453–2460.
412. Electron Transfer Cascade by Organic/Inorganic Ternary Composites of Porphyrin, Zinc Oxide Nanoparticles, and Reduced Graphene Oxide on a Tin Oxide Electrode that Exhibits Efficient Photocurrent Generation Hayashi, H.; Lightcap, I. V.; Tsujimoto, M.; Takano, M.; Umeyama, T.; Kamat, P. V.; Imahori, H. J. Am. Chem. Soc. 2011, 133. 7684–7687.
407. Graphene-based Composites for Electrochemical Energy Storage Radich, J. G.; McGinn, P. J.; Kamat, P. V. Interface 2011, Spring Issue, 63-66.
404. Graphene-based Nanoassemblies for Energy Conversion Kamat, P. V. J. Phys. Chem. Lett. 2011, 2, 242-251. (Perspective article)
